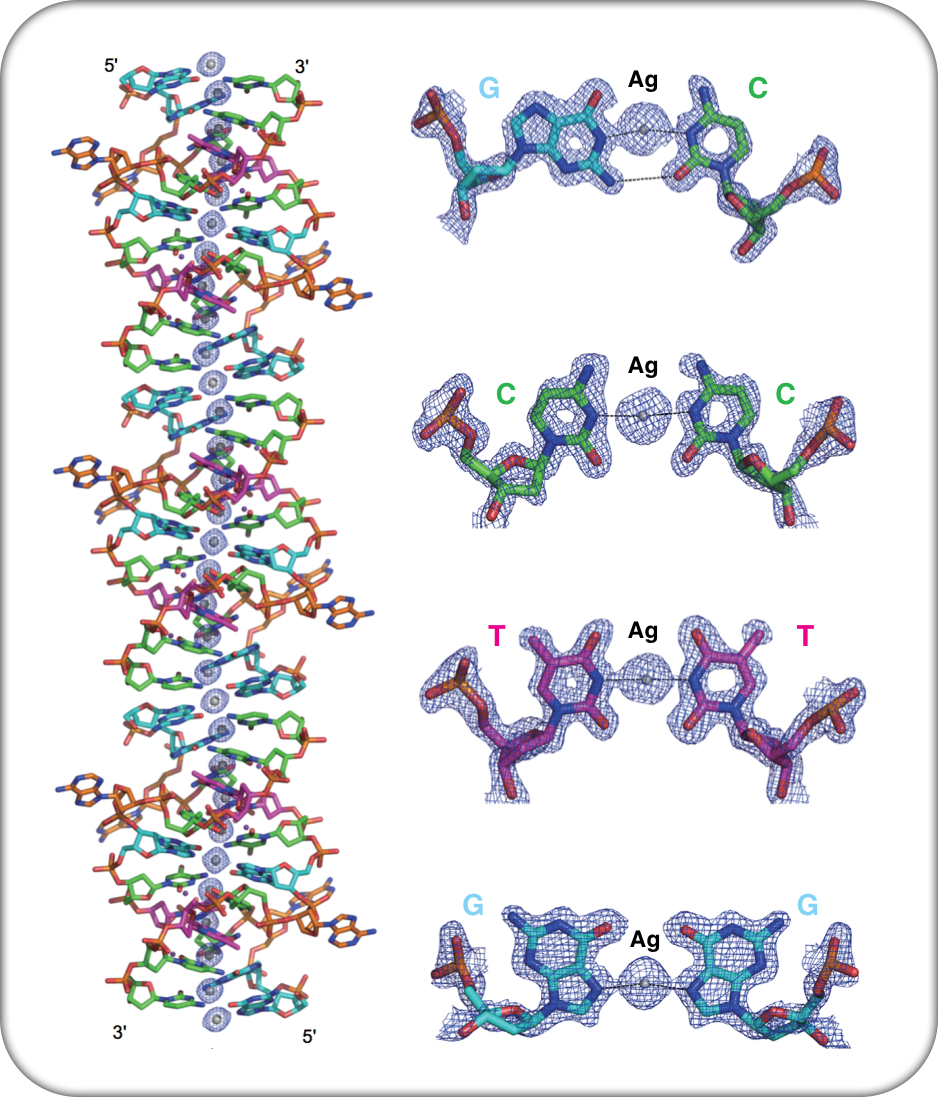
A DNA double helix, stabilized by silver ions forms a long, thin nanowire.
We are surrounded by so many electronic gadgets every day. Everything from your favorite iPod to that miniaturized digital watch contain microchips and very thin wires and circuits. Scientists are always on the exploration for materials on the order of nanometers so they can use it to build futuristic devices, where a full computer can be jam-packed into a needle head. In a recent study published in Nature Chemistry, researchers report a silver nanowire created using a DNA template. In their study, the DNA double helix contained non-canonical base pairs (i.e. base pairing except the traditional A:T/G:C) stabilized by silver ions. The "metallo base pairs" have provided a strategy to create silver/DNA hybrid nanowires, much longer than any nanowire previously reported. This study adds to the development of molecular electronics, one of the current focuses of bottom-up nanotechnology.
In recent years, DNA has been used for drug delivery, biosensing, molecular computation, and in the development of nanoelectronics. DNA, in its natural form, lacks electrical conductance and is unsuitable for applications in nanoelectronics. So scientists use DNA as a template for creating metallic nanowires, as a means to increase its electrical conductance while creating thin nanowires (on the order of a few nanometers). Up until this study, nanowires based on DNA duplexes with metallo base pairs were short, with around ten contiguous metallobase pairs. It remained a challenge to create long nanowires containing such metal ions.
Tanaka and coworkers now address this challenge by creating DNA nanowires in which the duplex contains metal-mediated base pairs. In their study, they used a DNA dodecamer (a DNA strand with 12 nucleotides) and crystallized it in the presence of silver nitrate. The crystal contained DNA duplex units composed only of silver-mediated base pairs. They used X-ray diffraction to study the crystal structure of metal-containing DNA duplexes: the duplexes in this crystal was much similar to the traditional, and well known, B-form conformation, except that the base pairs were not A:T or G:C. What they found was four types of silver mediated base pairing: C–Ag–C, G–Ag–G, G–Ag–C and T–Ag–T. Tanaka's group had earlier reported the silver-mediated C-Ag-C metallo base pair, but the other three were observed for the first time. One other observation was that the duplex was stabilized by exclusive silver-mediated base pairing causing the adenine residues in the dodecamer to bulge out from the duplex. In the crystal, the DNA duplexes were connected end-to-end by the guanine overhangs that formed a G-Ag-G base pair between the duplexes. This "continuous" duplex thus is a long silver nanowire within the DNA duplex. The aspect ratio of these nanowires are in the order of 50000 (diameter of the largest crystals was 0.1 mm and width of the continuous duplex DNA is 2 nm) which is larger than most reported nanowires. We have to wait to see if these nanowires can actually support electron transport either in solution or in crystallo.
The metal-mediated base pairs also lead to interesting options for DNA nanotechnology and molecular circuits. One, it opens up more sequence design options: we might be able to use metal-mediated non-canonical base pairs. Second, the metallized DNA itself imparts additional characteristics on to such DNA nanostructures (electrical and optical properties, for example). The inter-duplex connection in this study is also provided by metal mediate base pairing. DNA arrays depend on base pair hybridization, and many motifs are designed with sticky ends as structural glue. So one could envision a “metallo sticky end” by using silver-mediated interactions between connecting units. With developments in DNA nanotechnology and the new results of metal-mediated base pairs, newer possibilities are awaiting the field of nanoelectronics.
Discussion paper: J Kondo, et al, A metallo-DNA nanowire with uninterrupted one-dimensional silver array. Nat. Chem. doi:10.1038/nchem.2808 (2017).
This post is based on my highlight in ChemBioChem: AR Chandrasekaran, DNA arrays with a silver lining. ChemBioChem. doi:10.1002/cbic.201700367 (2017).